Season’s Greetings and welcome to our second newsletter! We hope you enjoy this edition.
This has truly been a foundational year for NFX UK in which we have firmly established our presence within the industry. Thank you for your support this year – we look forward to deepening our collaboration and driving meaningful initiatives together in 2026.
Remember, we are always working to enhance our newsletters. Let us know which topics, insights or updates you would like us to include in future editions – we welcome your ideas! Contact us at hello@nfxuk.co.uk
In the spirit of the season, we wish you a joyful, restful and prosperous festive period.
Included in this edition:
- NFX: Making connections
- Regulatory insights
- Novel Food sector news
- Cell-cultivated product guidance
- Additional FSA updates
- NFX website update
NFX: Making connections
The NFX UK team has had a busy few months, taking part in a range of Novel Foods events across the UK and Europe. These included the IFST Autumn Conference, Business Connect – Food Sector Group, the BioRefinery Network Annual Conference, EIT Next Bite and the Regulating the Future of Food Conference.
Throughout these events, we engaged with the community, shared the NFX UK mission and explored new opportunities for collaboration. A summary of some insights from the events follow.



Regulatory insights
- The UK’s Food Standards Agency (FSA) has been active in promoting its Innovation Research Programme (IRP), which focuses primarily on products developed through precision fermentation. They also highlighted the programme’s and the support available through the Business Support Service
- NFX UK presented at the recent Westminster Nutrition Forum, where it was encouraging to see our partners, experts and members contributing to the discussion on the future of Novel Foods in the UK. The message was clear: innovation and regulation must work hand in hand to balance safety with the rapid adoption of emerging food technologies, allowing the UK to fully capitalise on its strong scientific base for economic growth
- Several sessions across different events shared practical tips for preparing Novel Food dossiers globally. Key advice included:
- Identify your target market early and ensure commercial viability within that market
- Aim to build a ‘global data package’ that can support multiple regulatory submissions
- If you generate more data than required, include only the relevant information in your dossier to avoid slowing down review and approval.
- Clearly identify confidential aspects in the dossier such as process details
- Engage in early discussions with regulators (where available) to avoid avoidable gaps in submissions (e.g., missing antifoam analysis because its use in the process was not initially recognised as requiring data)
While dossier preparation is undeniably challenging and regulatory timelines are often criticised, regulators worldwide continue to emphasise that dossier quality remains one of the main factors affecting approval timelines.
Novel Food sector news
Industry news
Emerging Novel Foods pipeline
There was widespread celebration across events following notable recent milestones in the sector, including PARIMA’s approval in Singapore for its cultivated chicken and Clean Food Group’s investment in a UK scale-up facility. These events provide strong indicators of the emerging pipeline for Novel Food products
Global news
US FDA shutdown
From 1 October – 12 November 2025, the US FDA underwent a 43-day shutdown of non-essential activities due to a lapse in federal appropriations. During this period, pre-market reviews for Novel Food and animal feed ingredients were suspended, including GRAS notices, food additive petitions and new ingredient notifications. With funding now restored, the FDA faces the significant challenge of working through the backlog of submissions accumulated during the shutdown
All of these insights are informing NFX UK’s strategic direction, shaping how we support innovators and engage with policymakers to build a more resilient and collaborative ecosystem.
Keen to stay in the know?
Sign-up to join our NFX Community and follow our NFX UK LinkedIn page to see the latest FSA updates. Want the latest developments and insights delivered straight to your inbox?
Cell-cultivated product guidance
The FSA and FSS have published the UK’s first set of bespoke regulatory guidance for Cell-Cultivated Products (CCPs). Two key documents have been issued:
Cell-cultivated products: Classification and HACCP principles
This guidance is designed to help businesses understand and correctly apply hygiene regulation requirements when producing CCPs.
It confirms that CCPs produced using animal cells, often referred to as ‘lab-grown meat’, are classified as products of animal origin. As a result, producers must comply with all existing food safety regulations that apply to such products.
A major focus of the guidance is the mandatory use of Hazard Analysis and Critical Control Points (HACCP) principles across the entire production process to ensure robust food safety management.
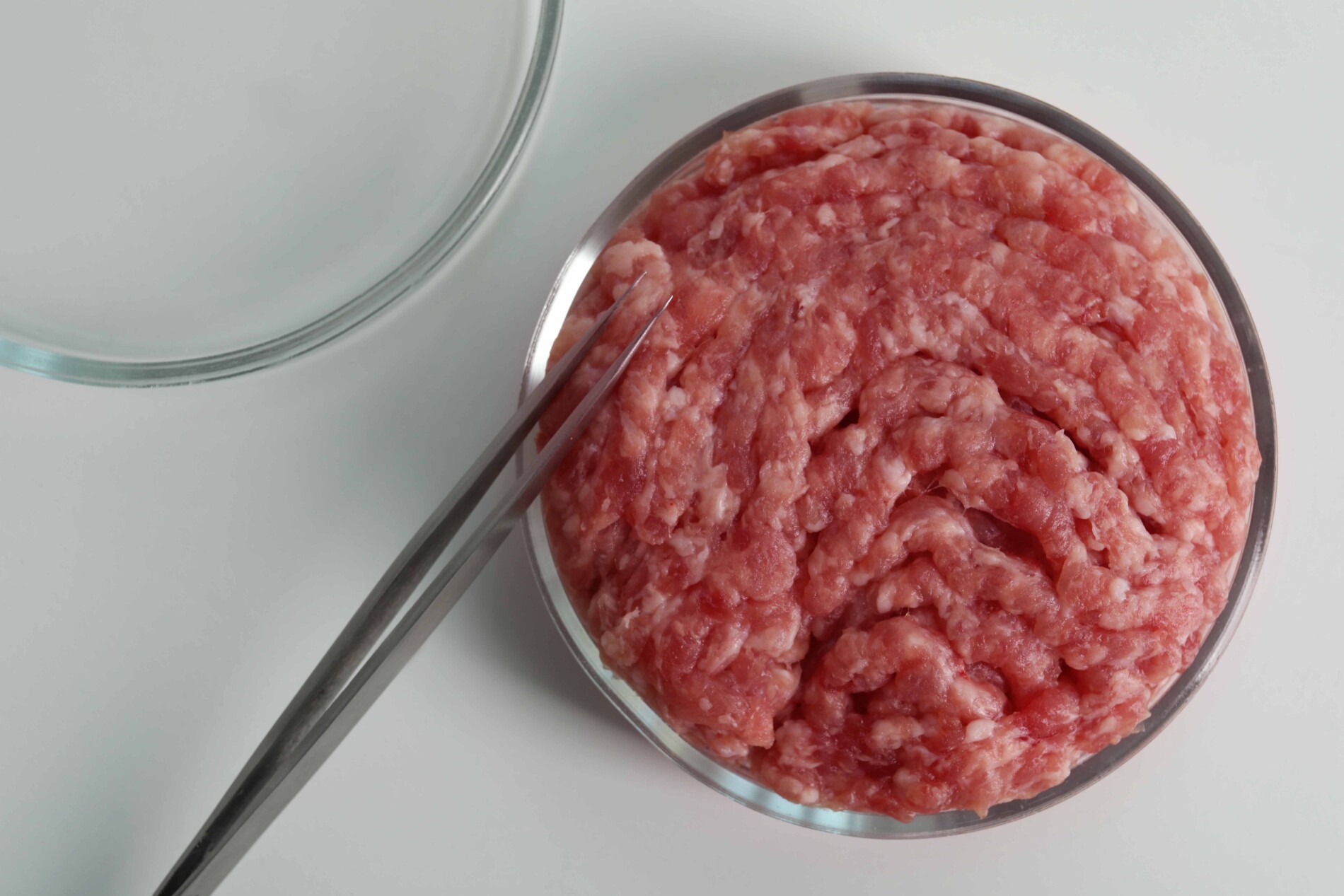
Supplementary guidance for applicants: Allergenicity and nutrition
The second guidance sets out the requirements for assessing allergenicity and nutritional quality when applying for market authorisation of CCPs as Novel Foods in Great Britain. These evaluations are essential for successful market entry.
Allergenicity assessments
The guidance details how applicants should assess potential allergens in CCPs, including consideration of how the novel production method may affect allergenic potential. This is critical for consumer safety and accurate labelling.
Nutritional quality assessments
It also describes the methodology and criteria used to evaluate the nutritional quality of CCPs as part of the overall Novel Foods approval process. This ensures products meet appropriate nutritional standards and provide transparent information to consumers.
This initial release forms part of the wider Cell-Cultivated Sandbox Programme, with additional regulatory and technical guidance expected over the coming year. Together, these publications aim to provide greater clarity and certainty for businesses operating in this rapidly evolving sector.
Additional FSA updates
Guidance for Food Business Operators (FBOs) on researching novelty status of food
On 19 November, the Food Standards Agency (FSA) issued an important update to its guidance for Food Business Operators (FBOs) on determining the Novel Food status of products in Great Britain.
The updated guidance provides clearer direction and now includes direct links to key resources that FBOs can use to conduct their own preliminary assessments of whether a product may be considered novel under the Novel Food Regulation. This enables businesses to carry out informed research early in the development process.

The key change from the FSA is that they will no longer provide informal advice on whether a product is novel. Previously, businesses could seek unofficial feedback, but going forward, they will be directed to use the official guidance to conduct their own research.
The formal process for legally verifying Novel Food status via an Article 4 consultation remains unchanged and continues to be the statutory route for businesses requiring an official determination.
We encourage all food businesses to familiarise themselves with this updated guidance to ensure on-going compliance and the confidence to navigate the Novel Food regulatory landscape. The NFX UK Knowledge Hub and the Expert Directory can offer rich resources for FBOs to navigate this space.
Taste trials guidance
The FSA has also released updated guidance on conducting taste trials involving Novel Foods or foods produced through novel processes, with the latest revision published on 24 October.
Novel Foods must be authorised before being placed on the GB market. ‘Placing on the market’ is defined broadly and may include trade samples. Taste trials intended for research and development may be permitted for unauthorised Novel Foods. However, trials conducted for promotional purposes may be considered the unlawful placing on the market of an unauthorised Novel Food.
This updated guidance aligns with the Advisory Committee on Novel Foods and Processes (ACNFP) principles and aims to support companies in designing safe, ethical and compliant taste trials. It covers:
- Key definitions
- Referral protocol
- Risk assessment
- Ethics committee approval
- Record keeping
- GMO notification, where applicable
The FSA and FSS advise companies to carry out thorough participant safety checks before running any taste trial. When using the Novel Foods authorisation guidance on the FSA website, several areas are strongly recommended for inclusion in a robust risk assessment.
Market authorisation innovation research programme (IRP) update

Following the launch of the FSA and FSS Innovation Research Programme (IRP) – an initiative designed to strengthen regulatory readiness for emerging food technologies, particularly precision fermentation – the NFX UK team recently held a productive in-person meeting with members of the IRP team.
During the meeting, we agreed several collaborative actions and established a joint roadmap that will support and strengthen the Novel Foods space. This partnership will help ensure that industry perspectives are clearly represented and communicated.
The IRP offers valuable resources, including:
- A guidance hub providing regulatory advice on Novel Food technologies, with a focus on precision fermentation
- A dedicated business support service offering expert assistance with applications
Please watch out for upcoming surveys and updates, which we will share with our network.
NFX website update
The NFX UK Network continues its rapid growth, proudly reaching 200+ registered members, including 70+ experts now featured in our directory. Through ongoing collaboration with key regulatory bodies such as the FSA and FSS, we remain committed to shaping the future of food systems in the UK. The network is driving forward-thinking solutions for a greener, healthier food industry through collaboration and engagement.
To better support this work, we are evolving our digital platform. Get ready for the next evolution of the NFX UK website – Phase III is coming soon, featuring:
- Newly curated resources by the NFX UK team and collaborators, including the next layer of our comprehensive Novel Foods Roadmap
- An Organisational Directory to help you connect easily with organisations offering relevant services, technologies and expertise in the Novel Food sector
Unlock expert support: Access our Expert Directory
Looking for specialised guidance? Our Expert Directory connects you with leading professionals in key areas such as nutritional science, biotechnology, regulatory, food safety and analysis and testing, who can help accelerate your innovation journey.
How to access the directory:
- Sign up for free or log in to your NFX UK account on our website
- Navigate to the ‘Knowledge Hub’ section
- Click on ‘Expert Directory’
- Browse the directory by area of expertise or search for specific keywords
Coming soon from NFX UK
- Webinars and training courses
- Continued development of relationship with FSA and FSS and more regulatory updates
- Investor workshop to help investors learn more about the Novel Food regulatory landscape

Join our community
Sign up for exclusive access to the NFX UK Knowledge Hub.
Join our Novel Foods Network today for free and get insider information regulation, industry knowledge and access to the experts. Becoming a member is easy, all you need to do is fill out the form. If you have any questions, visit our FAQs page or Contact Us.
If you are already a member, log in here.



